DermNet provides Google Translate, a free machine translation service. Note that this may not provide an exact translation in all languages
Home Wound healing Normal wound healing CME
Wound healing
Normal wound healing
Created 2009.
Learning objectives
- Describe epidermal and dermal wound repair
- Name cells and cytokines involved in wound healing
Introduction
Skin generally heals rapidly to restore barrier function after injuries such as cuts, abrasions, thermal burns, puncture wounds and blisters.
The severity of a wound depends on its surface area and depth. Partial thickness wounds can recruit new keratinocytes from adjacent adnexal tissue and can regenerate much more rapidly than full thickness wounds of the same surface area where regeneration of epithelium arises only from keratinocytes on the wound edges. Damage to the dermis can be repaired but results in altered tissue i.e. a scar. However, fetal wounds heal by regeneration without fibrosis so do not result in scars.
Injury results in epidermal and dermal repair:
- Haemostasis
- Inflammation
- Proliferation
- Repair
Haemostasis creates a protective wound scab beneath which cell migration and movement of the wound edges can occur.
Inflammation brings nutrients to the area of the wound, removes debris and bacteria, and provides chemical stimuli for wound repair.
Repair begins immediately after wounding and proceeds rapidly through the processes of epithelialisation, fibroplasia, and capillary proliferation into the healing area.
Wounds can be classified as either acute or chronic. Acute wounds are sometimes defined as those that follow the normal phases of healing; they are expected to show signs of healing in less than 4 weeks and include postoperative wounds. Chronic wounds are those that persist for longer than 4 weeks and are often of complex poorly understood origin.
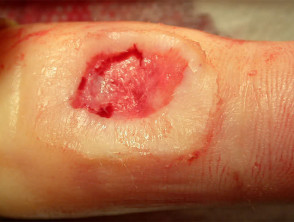
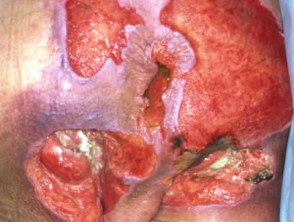
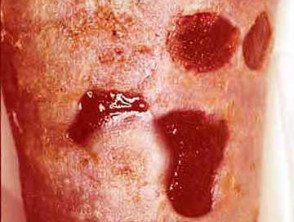
Wounds
Epidermal repair
- Vasoconstriction to reduce blood loss.
- Blood clotting: plasma fibrinogen is cleaved to form strands of fibrin, which, with platelets, are deposited on the wound surface.
- Fibrin binds fibronectin, which in turn binds keratinocytes and enhances the migration and proliferation of keratinocytes.
- The proliferation and migration of keratinocytes from the edges of the wound and adnexae results in a single layer of epithelium with an immature basement membrane zone.
- Restoration of basement membrane laminin, then type IV collagen and then anchoring fibrils occurs quickly over an intact dermis but is slow over granulation tissue.
- Restoration of stratified epithelium. When the wound surface is completely covered by epithelial cells, the scab sloughs off and the epidermis begins to keratinise.
Dermal repair
- Dermal repair is partly dependent on re-epithelialisation to maintain a moist clean environment.
- Chemotactic factors are released from platelets and injured tissues to attract neutrophils (first 3 days) and monocytes, which remove foreign material, bacteria and cellular debris by phagocytosis.
- Complement activation (C3a, C5a) results in vasodilation and inflammation.
- Nitric acid regulates various cellular activities of the inflammatory and proliferative phases of wound healing.
- Growth factors are released by platelets:
- Platelet derived growth factor
- Transforming growth factor-b.
- Macrophages initiate and maintain proliferative phase of dermal wound healing:
- Platelet derived growth factor
- Transforming growth factor-b
- Interleukin-1
- Fibroblast growth factor.
- Granulation tissue is evident at about 5 days due to proliferating macrophages, fibroblasts, smooth muscle and endothelial cells.
- Wound contraction is mediated by myofibroblasts, which contain abundant actin filaments.
- Fibroblasts produce fibronectin, which acts as a scaffold first for the fibroblasts and then for collagen fibrils (first type III and later type I collagen).
- Fibroblasts also produce hyaluronic acid and later proteoglycans (chondroitin-4-sulphate and dermatan sulphate), which absorb water and occupy the bulk of the extracellular matrix.
- The collagen matrix slowly transforms granulation tissue into scar tissue.
- Excessive fibroblast proliferation and collagen synthesis are inhibited by interferon b and g.
- Remodelling of the collagen matrix may continue for months or years, with collagenase degrading the collagen and fibroblasts creating more and stronger parallel fibres of collagen. Despite this, the scar is rarely as strong as the tissue it replaced. It is at maximum strength 12 weeks after the injury.
- As vascularization decreases, the colour of the wound fades.
- Damaged hair follicles, sweat glands and melanocytes may not be replaced so the scar has a smooth uniform surface.
Moist wound healing
A moist environment is beneficial compared with a dry wound. Moist wounds:
- Prevent further tissue loss by desiccation
- Promote activity of lytic enzymes
- Accelerate the cellular phase of collagen matrix deposition
- Facilitate keratinocyte migration.
Activity
Describe typical acute and chronic wounds seen in your clinic recently.
Related information
References
- A Bayat, D A McGrouther, M W J Ferguson. Skin scarring. BMJ 2003; 326: 88-92
On DermNet NZ
Information for patients
Other websites
- Medscape Reference:
- Wound Bed Preparation: The Science Behind the Removal of Barriers to Healing
- Wound care information network
- Wound care manual and clinical guideline for nurses: AusMed
- Merck Manual Professional: Lacerations: Physiology
Books about skin diseases
- Ruth Bryant Acute and Chronic Wounds
- Clinical Guide to Wound Care
See the DermNet NZ bookstore
Sign up to the newsletter
© 2024 DermNet.
DermNet does not provide an online consultation service. If you have any concerns with your skin or its treatment, see a dermatologist for advice.