Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Author: Dr Julie Smith MBChB FRACP, Dept of Dermatology Greenlane Hospital, Auckland, New Zealand, 2004; minor update Feb 2025.
Introduction
Uses
How it works
Safety
How to use
Side effects
Drug interactions
Ivermectin is a macrocyclic lactone medicine used to treat parasitic infections. In New Zealand, ivermectin is available in 3 mg tablets (Stromectol™). Ivermectin cream (Soolantra™) was approved by the FDA in December 2014 but is not yet readily available.
Ivermectin is effective against infections with:
Ivermectin has been used extensively as part of the World Health Organisation's Onchocerciasis Eradication Program. It has FDA approval for the treatment of strongyloides and onchocerciasis in the United States, other use is termed ‘off label’.
In treatment of scabies its use may be appropriate in selected cases where topical therapy is impractical or has failed and is particularly useful in cases of crusted scabies (also called ‘Norwegian’ scabies).
Ivermectin can also be used to control demodicosis. Ivermectin cream has been shown to reduce papulopustular rosacea, which is thought to be due to its effects on demodex mites and its anti-inflammatory action.
A 2021 Cochrane Review has not found sufficient evidence to support the use of ivermectin for preventing or treating COVID-19.
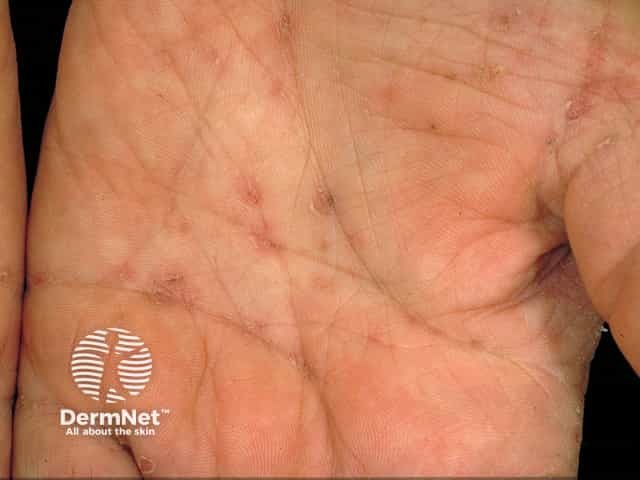
Burrows
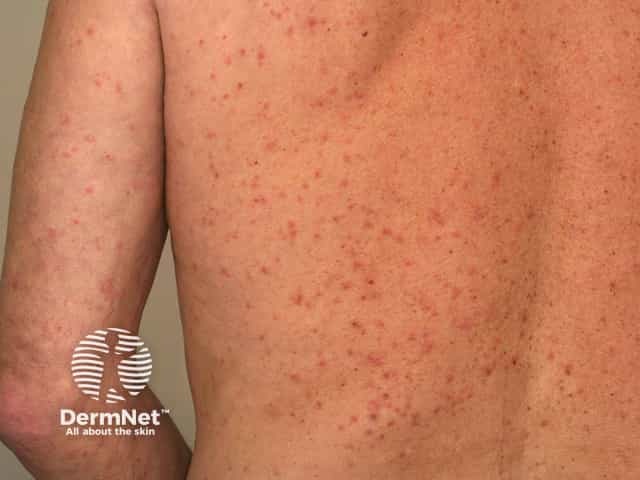
Scabies hypersensitivity rash
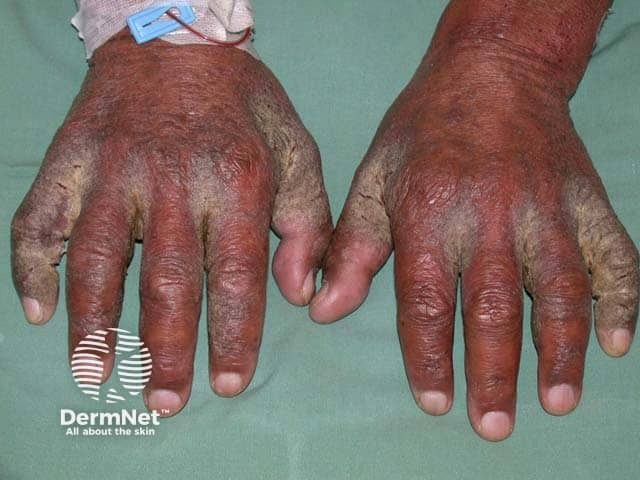
Crusted scabies
Ivermectin stimulates excessive release of neurotransmitters in the peripheral nervous system of parasites. It is thought to work by paralysing the parasite or inactivating the parasite gut. In humans the neurotransmitters acted on by ivermectin are in the brain. A protective barrier, called the blood-brain barrier, blocks ivermectin from reaching the human brain.
Oral ivermectin appears to be a safe medication.
Ivermectin is primarily metabolised in the liver by CYP450-3A47 and has a plasma half life of 16 hours. It is almost exclusively excreted in faeces with minimal clearance by the kidneys. It therefore does not require dose adjustment for people with renal failure. There have been some reports of a mild anti-coagulation effect. However, this is usually not significant enough to alter coagulation parameters such as the prothrombin ratio.
Ivermectin is taken as a single dose with a glass of water. The tablets can be crushed, for example to be given via a percutaneous endoscopic gastrostomy (PEG) tube. It is rapidly absorbed from the gut and metabolised in the liver.
| Infestation | Dose regime |
|---|---|
| Onchocerciasis | Single dose of 150mcg/kg (may be repeated every 3 to 12 months) |
| Strongyloidiasis | Single dose of 200mcg/kg |
| Scabies | Dose of 200mcg/kg repeated in 7 to 14 days |
| Cutaneous larva migrans | Single dose of 200mcg/kg |
Side effects of oral ivermectin are rare and usually minor. These include:
According to French dermatology guidance (2024), ivermectin can be used in the second trimester of pregnancy and onwards; it can also be given to breastfeeding women. Ivermectin appears to be safe in children over the age of 2 years. Ivermectin may be an option as second-line therapy in children over the age of 2 months, however administration of the available formulation may be a barrier.
Ivermectin cream can irritate and cause a burning sensation on treated skin.
No significant drug interactions with ivermectin are recognised.
New Zealand approved datasheets are the official source of information for these prescription medicines, including approved uses and risk information. Check the individual New Zealand datasheet on the Medsafe website.