Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Autoimmune/autoinflammatory
Author: Anoma Ranaweera, Medical writer, 2011. DermNet Editor in Chief: Adjunct A/Prof Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand. Reviewed and revised April 2019.
Introduction
How it works
Dosage
Adverse events
Precautions
Vaccinations
Drug interactions
Use of ustenkinumab
Monitoring
Ustekinumab (brand name STELARA™) is a biological treatment used to treat moderate to severe psoriasis. It is a human monoclonal antibody that antagonises interleukin-12 (IL-12) and IL-23. Good to excellent responses are seen in more than two-thirds of patients with chronic plaque psoriasis treated with ustekinumab.
Ustekinumab has been shown in small studies to be useful in other forms of psoriasis, including nail psoriasis, erythrodermic psoriasis, generalised pustular psoriasis and palmoplantar pustulosis. It is also used to treat psoriatic arthritis and Crohn disease.
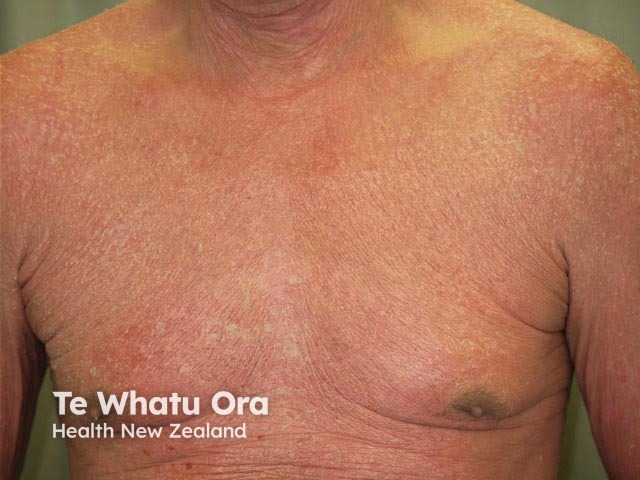
Erythrodermic psoriasis before ustekinumab
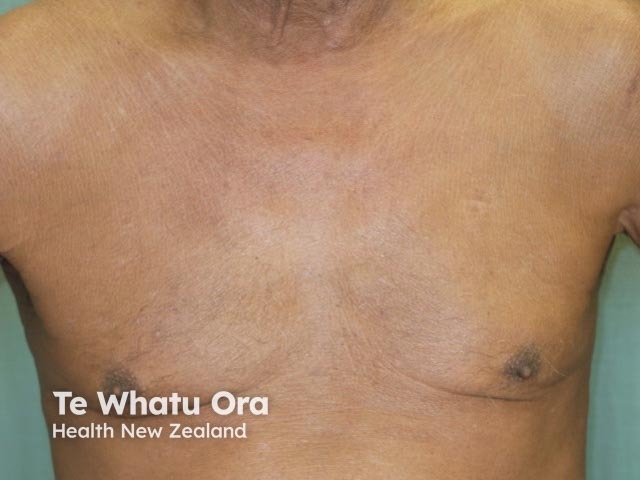
Erythrodermic psoriasis 2 weeks after first dose of ustekinumab
Psoriasis is caused by an increase in the production of T-cells and is influenced by cytokines, the chemical messengers produced by cells. Certain cytokines cause skin cells to grow rapidly, producing plaques of psoriasis. Ustekinumab is a monoclonal antibody that targets the p40 subunit of the cytokines IL-12 and IL-23, preventing them from binding and activating T-lymphocytes.
Ustekinumab is given by subcutaneous injection. The second dose is given 4 weeks after the first injection, and further doses are delivered every 12 weeks. It reaches its peak effect at around 28 weeks.
To date, adverse infections are consistent with that seen with other biologics.
Immunisation status should be reviewed prior to starting ustekinumab. If necessary, vaccines should be updated prior to treatment with a biologic agent. Annual influenza vaccination is recommended.
As they may induce illness in immunodeficient individuals, live vaccines should not be used during treatment with ustekinumab. Currently, available live attenuated viral vaccines include measles, mumps, rubella, varicella, yellow fever, influenza (intranasal vaccine) and the oral polio vaccine. Live attenuated bacterial vaccines include BCG and oral typhoid vaccine.
Read more about immunisation in immunosuppressed dermatology patients.
There are no studies of ustekinumab in pregnant women. Ustekinumab should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Caution should be exercised when ustekinumab is administered to a nursing woman. The unknown risks to the infant from gastrointestinal or systemic exposure to ustekinumab should be weighed against the known benefits of breastfeeding.
A study has shown the safety and effectiveness of ustekinumab in children aged 12 to 17 years to be similar to that in adults.
No pharmacokinetic data are available in patients with liver or kidney disease treated with ustekinumab.
It is recommended that patients on biologic medications have routine blood tests every 6 months or so, including full blood count and liver function tests. TB testing should be repeated from time to time.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).