Introduction
In December 2016, the US Food and Drug Administration (FDA) approved crisaborole topical ointment, 2%, to treat mild to moderate atopic dermatitis in patients 2 years of age and older. The age limit in the US has subsequently been changed to include patients from 3 months of age.
Crisaborole 2% ointment has subsequently been approved for use in Australia and New Zealand (2019) on prescription for patients from 2 years of age with mild to moderate atopic dermatitis.
Crisaborole is a boron-based phosphodiesterase 4 inhibitor (PDE-4). It is a non-steroidal topical monotherapy that inhibits the phosphodiesterase (PDE)-4 enzyme in the skin. Overactive PDE-4 has been shown to contribute to the signs and symptoms of atopic dermatitis.
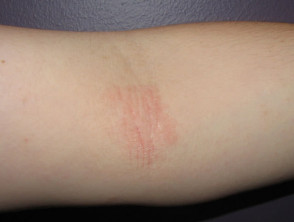
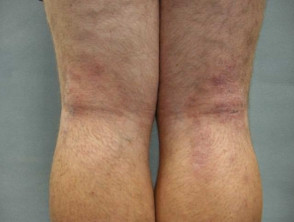
Atopic dermatitis
How does crisaborole work?
- The specific mechanism(s) by which crisaborole exerts its therapeutic action is not well defined.
- Atopic dermatitis is characterized by type 2 helper T (Th2) cell-driven inflammation.
- Crisaborole is an inhibitor of the enzyme PDE-4.
- PDE-4 inhibition results in increased intracellular cyclic adenosine monophosphate (cAMP) levels.
- Crisaborole modifies inflammation in atopic dermatitis by inhibiting the degradation of cAMP by PDE4, resulting in downstream modification of nuclear factor-kB and T-cell signalling pathways.
Dosage and administration
- Crisaborole is for topical use only and not for ophthalmic, oral, or intravaginal use.
- It is applied as a thin layer over the affected area(s) twice daily.
- Shown to be effective, well tolerated, and safe when used as maintenance treatment for up to 52 weeks to control eczema.
- Inactive ingredients in the ointment include white petrolatum, propylene glycol, mono- and diglycerides, paraffin, butylated hydroxytoluene, and edetate calcium disodium.
Use in specific populations
Pregnancy
- There is no available data for use of crisaborole in pregnant women to inform of drug-associated risk for major birth defects and miscarriage.
- No adverse developmental effects were observed in pregnant rabbits with oral administration of crisaborole.
- See DermNet page on the safety of medicines taken during pregnancy.
Lactation
- There is no information on the presence of crisaborole in human milk or the effects of the drug on the breastfed infant after topical application in women who are breastfeeding.
- Crisaborole is systemically absorbed.
- The risk-benefit potential should be considered when prescribing crisaborole to the mother.
- See DermNet page on lactation and medications used in dermatology.
Paediatric use
- The safety and effectiveness of crisaborole have been established in paediatric patients age 2 years and older.
- The safety and effectiveness of crisaborole in children below the age of 2 years have not been established.
Geriatric use
- Clinical studies of crisaborole did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects.
What are the possible adverse effects of crisaborole?
- The most common side effect of crisaborole is application site pain, such as burning or stinging.
- Crisaborole may cause hypersensitivity reactions at the application site or at a distant site.
- These can be serious and may include hives (contact urticaria), itching, swelling, and erythema.
- If signs and symptoms of hypersensitivity occur, crisaborole should be discontinued immediately and appropriate therapy initiated.
- Crisaborole is contraindicated in patients with known hypersensitivity to crisaborole or any component of the formulation.
Tell me more about Crisaborole?
- Crisaborole is a first-in-class, nonsteroidal, topical anti-inflammatory, PDE inhibitor approved for the treatment of mild to moderate atopic dermatitis.
- Crisaborole represents a promising, nonsteroidal topical treatment to improve management of atopic dermatitis.
- Significant improvement in disease severity, pruritus intensity, and all other assessed signs and symptoms of atopic dermatitis indicate that crisaborole may target the underlying pathogenesis of the disease.
- The novel boron chemistry of crisaborole enables synthesis of a low-molecular-weight compound (251 d) that facilitates effective penetration through human skin.
Does crisbarole help other skin conditions?
Crisaborole has been reported to be of benefit (but not yet approved) for intertriginous/flexural psoriasis and facial psoriasis, and chronic irritant contact dermatitis.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).