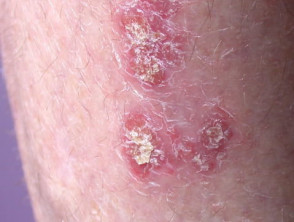
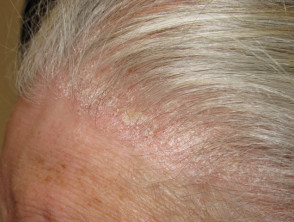
What is calcipotriol/betamethasone dipropionate ointment and gel?
Calcipotriol/betamethasone dipropionate ointment and gel are topical medications for the treatment of plaque psoriasis [1–3]. The brand names of calcipotriol/betamethasone dipropionate are Daivobet® 50/500 ointment and Daivobet® 50/500 gel. Calcipotriol is called calcipotriene in some countries. An alternative brand name for the combination is Dovobet®.
Calcipotriol/betamethasone dipropionate ointment and gel are available in New Zealand with a doctor’s prescription and are fully funded by PHARMAC. The New Zealand marketing authorisation for Daivobet is held by LEO Pharma.
Psoriasis suitable for treatment with calcipotriol/betamethasone dipropionate
Key phase III trials in scalp psoriasis with calcipotriol/betamethasone diproprionate gel
In an 8-week, multicentre, randomised, double-blind study, patients with scalp psoriasis were randomised to treatment with the two-compound scalp formulation (calcipotriol 50 mcg/g plus betamethasone 0.5 mg/g, as dipropionate) (n = 541), betamethasone 0.5 mg/g (as dipropionate) in the same vehicle (n = 556), calcipotriol 50 mcg/g in the same vehicle (n = 272) or vehicle alone (n = 136) [4].
- Primary response criteria were controlled disease at week 8, defined as ‘clear’ or ‘minimal disease’ for patients with moderate disease at baseline, or ‘clear’ for patients with mild disease at baseline according to International Global Assessment (IGA) criteria.
- More patients achieved 'absent' or 'very mild' disease at week 8 with the two-compound scalp formulation (71.2%) compared with betamethasone dipropionate in the same vehicle (64.0%, p = 0.011), calcipotriol in the same vehicle (36.8%, p < 0.0001), or the vehicle (22.8%, p < 0.0001).
Long-term study
A long-term (52-week) randomised, double-blind study in 869 patients with moderate-to-severe scalp psoriasis investigated the efficacy of the two-compound formulation (calcipotriol 50 mcg/g plus betamethasone dipropionate 0.5 mg/g) for scalp psoriasis in comparison to calcipotriol [5].
- Patients were randomised to either a two-compound scalp formulation (n = 429) or calcipotriol (n = 440).
- The primary response criteria were the incidences of both adverse drug reactions (ADRs) of any type and adverse events of concern associated with long-term topical corticosteroid use on the scalp.
- Secondary criteria included efficacy of the treatment based on the Investigator's Global Assessment of disease severity (‘absence of disease’, ‘very mild disease’, ‘mild disease’, ‘moderate disease’, ‘severe disease’, ‘very severe disease’) and patient ratings of the treatments efficacy (‘satisfactory’, ‘not satisfactory’).
- The percentage of patients reporting adverse drug reactions (ADR) was significantly lower in the two-compound group compared with the calcipotriol group [17.2% (n = 72/419) vs. 29.5% (n = 127/431); odds ratio (OR): 0.5, 95% confidence interval (CI): 0.36–0.69, p < 0.001.
- The proportion of patients with ADRs who used the medication as needed throughout the initial 6 months of participation was lower in the two-compound group compared with the calcipotriol group [11.4% (n = 40/350) vs. 22.4% (n = 66/295)]. This was also seen in patients who used the treatment for the entire 12-month period [15.7% (n = 44/281) vs. 26.4% (n = 62/235)].
- According to the patient assessments, the percentage of satisfactory visits per patient was significantly in favour of the two-compound scalp formulation (p < 0.001). A total of 76.2% of patients assessed the treatment response as satisfactory at every visit in the two-compound group versus 50.2% in the calcipotriol.
According to the Investigator's Global Assessment of disease severity, the number of patients who reached ‘satisfactorily controlled disease’ (‘absence of disease’, ‘mild disease’ or ‘very mild disease’) was higher in the two-compound group than in the calcipotriol group.
- The median number of visits per patient with ‘satisfactorily controlled disease’ was superior in the two-compound group. Patients in this group were rated as having satisfactorily controlled disease in 92.3% of the assessments, which was significantly more than in the calcipotriol group (80.0%; p < 0.001).
Key phase III trials in body psoriasis
- The efficacy of once-daily use of calcipotriol/betamethasone dipropionate gel on non-scalp regions of the body was investigated in a randomised, double-blind, 8-week clinical study including 364 patients with chronic plaque psoriasis of the trunk or limbs [6].
- Comparators were betamethasone dipropionate in the gel vehicle, calcipotriol in the gel vehicle and the gel vehicle alone, all used once daily.
- Results of the primary response criterion showed that the percentage of patients whose disease was clear or very mild and who had at least a two-step improvement in the Investigator's Global Assessment (IGA) of disease severity at week 8, was significantly higher with calcipotriol plus betamethasone dipropionate (27.2%) than with betamethasone dipropionate (16.9%, p = 0.027), calcipotriol (11.4%, p = 0.006) or gel vehicle (0.0%, p < 0.001).
Calcipotriol/betamethasone effectiveness in real-life studies
Several large-scale, real-life studies have assessed the efficacy of calcipotriol/betamethasone gel in plaque psoriasis of the body and scalp. Besides the demonstration of antipsoriatic efficacy in rigidly controlled clinical trials, confirmation of effectiveness in the real-life setting where factors such as patient preference, satisfaction, and adherence to treatment become increasingly relevant.
- In the largest of these studies (the PSOriasis Treatment OPtimisation [PSO-TOP] study; n=1,795), 4 after 8 weeks treatment with calcipotriol/betamethasone topical gel, more than one-third of patients who had previously failed treatment with potent topical steroids had documented PGA (Patient’s Global Assessment) or PhGA (Physician’s Global Assessment) treatment success (lesions ‘clear’ 34.2% or ‘almost clear’ 36.5% ; p<0.0001 vs baseline) [8].
- Importantly, 48% of patients had received unsuccessful treatment with a potent topical steroid during the 8 weeks before calcipotriol/betamethasone.
- The Patient Preference Questionnaire revealed that calcipotriol/betamethasone gel was highly preferred by patients (> 85%) relative to previous topical therapy.
- Regardless of the previous treatment received, most patients ‘agreed’ or ‘strongly agreed’ that calcipotriol/betamethasone was more effective, easier to use and was associated with fewer adverse events and better tolerance than previous treatments.
Cost-effective analyses
Pharmacoeconomic analyses have shown calcipotriene/betamethasone dipropionate gel formulations are more cost-effective than other topical therapies for psoriasis [9].
Summary
- The gel formulation of calcipotriol/betamethasone provides an effective first-line option for the treatment of mild-to-moderate plaque psoriasis of both the body and scalp.
- The product is a once-daily formulation and provides faster and greater symptom relief than a potent topical steroid used alone.
- The formulation works in two ways to reduce inflammation and to treat the scaling associated with psoriasis.
- This product is fully funded in New Zealand.
Key phase III trials with calcipotriol/betamethasone dipropionate ointment
Trial 1
- An international, multicentre, prospective, randomised, double-blind, vehicle-controlled, parallel-group, 4-week study was performed in patients with chronic plaque psoriasis amenable to topical treatment [3].
- Patients were randomised to one of four treatment groups: combined calcipotriol/betamethasone dipropionate formulation once daily, combined formulation twice daily, calcipotriol twice daily, or vehicle twice daily.
- There was no statistically significant difference in the mean percentage change in the Psoriasis Area and Severity Index (PASI) from baseline to end of treatment between the two combined formulation groups, but the difference in PASI reduction was significantly higher in the combined formulation groups (68.6% once daily, 73.8% twice daily) than in both the twice-daily calcipotriol group (58.8%) and the vehicle group (26.6%).
- Safety data showed the frequency of adverse events to be less in the combined formulation groups than in both the calcipotriol group and the vehicle group.
- The proportion of patients with lesional/perilesional adverse reactions was less in the combined formulation groups and vehicle group than in the calcipotriol group (9.9% combined formulation once daily, 10.6% combined formulation twice daily, 19.8% calcipotriol, 12.5% vehicle).
- Overall, no statistically significant or clinically relevant difference in efficacy was seen between the combined formulation used once daily and twice daily. When compared to vehicle ointment or calcipotriol ointment alone, the combined formulation was shown to be clearly more efficacious.
Trial 2
- 972 patients with plaque psoriasis were randomised to one of three treatment regimens: Group 1, the two-compound calcipotriol/betamethasone dipropionate ointment once daily for 8 weeks followed by calcipotriol ointment once daily for 4 weeks; Group 2, the two-compound ointment once daily for 4 weeks followed by 8 weeks of treatment with calcipotriol ointment once daily on weekdays and the two-compound ointment once daily at weekends; and Group 3, calcipotriol ointment twice daily for 12 weeks [4].
- The efficacy was evaluated by PASI and investigators' global assessments of disease severity. The primary response criteria were percentage reduction in PASI and proportion of patients with absent/very mild disease according to the investigators' global assessments after 8 weeks of treatment.
- The mean reduction in PASI from baseline to the end of 8 weeks of treatment was 73.3% for Group 1, 68.2% for Group 2, and 64.1% for Group 3.
- The proportion of patients with absent/very mild disease at the end of 8 weeks of treatment was 55.3% for Group 1, 47.7% for Group 2, and 40.7% for Group 3.
- For both primary response criteria, Group 1 was statistically superior to Group 3 (p < 0.001), whereas Group 2 did not differ significantly from Group 3. The difference between Group 1 and Group 2 was statistically significant with regard to PASI but not with regard to the proportion of patients with absent/very mild disease.
- Patients receiving initial therapy with the two-compound product achieved the fastest treatment response, with the maximum treatment effect seen after 5 weeks. This effect was maintained with continued treatment with the two-compound product for up to 8 weeks.
- After 12 weeks of treatment, no significant differences were seen between the three groups with regard to reduction in PASI, whereas the proportion of patients with absent/very mild disease in Group 2 was superior to that in Group 3.
- Patients receiving therapy with the two-compound product experienced fewer lesional/perilesional adverse drug reactions than the calcipotriol-treated patients (P < 0.001): 10.9% in Group 1, 11.5% in Group 2, and 22.3% in Group 3.
Long-term study
The primary objective was to investigate the safety of two treatment regimens involving the use of the two-compound calcipotriol/betamethasone dipropionate product over 52 weeks [5].
Six hundred and thirty-four patients were randomised double-blind to treatment (once daily, when required) with either:
- 52 weeks of the two-compound product (two-compound group)
- 52 weeks of alternating 4-week periods of two-compound product and calcipotriol (alternating group)
- 4 weeks of the two-compound product followed by 48 weeks of calcipotriol (calcipotriol group). There was a trend towards the efficacy of the two-compound product used for up to 52 weeks being better than that of 4 weeks of the two-compound product followed by 48 weeks of calcipotriol.
Results
- Adverse drug reactions (ADRs) occurred in 45 (21.7%) patients in the two-compound group, 63 (29.6%) in the alternating group and 78 (37.9%) in the calcipotriol group. The odds ratio for an ADR in the two-compound group relative to the calcipotriol group was 0.46 (95% confidence interval 0.30-0.70; p < 0.001).
- ADRs of concern associated with long-term topical corticosteroid use occurred in 10 (4.8%) patients in the two-compound group, six (2.8%) in the alternating group and six (2.9%) in the calcipotriol group; those with the highest incidence were skin atrophy, occurring in four (1.9%), one (0.5%) and two (1.0%) patients, respectively, and folliculitis, in three (1.4%), one (0.5%) and no patients, respectively.
- Calcipotriol/betamethasone dipropionate ointment once daily lead to 2% fewer patients withdrawing (95% CI 0% to 4%, p = 0.03) due to adverse events and 11% fewer patients having at least one adverse event (95% CI 6% to 16%, p = 0.00001) than calcipotriol ointment once daily.
- Treatment with the two-compound product for up to 52 weeks appears to be safe and well-tolerated whether used on its own or alternating every 4 weeks with calcipotriol treatment.
Calcipotriol/betamethasone dipropionate ointment compared with calcipotriol and betamethasone alone
- 1106 patients with psoriasis were randomised to twice daily to double-blind treatment with betamethasone/calcipotriol ointment, betamethasone dipropionate alone, or calcipotriol for 4 weeks. Patients then received twice daily calcipotriol, unblinded, for a further 4 weeks [6].
- The mean percentage change in PASI at the end of the double-blind phase was -74.4 (combination group), -61.3 (betamethasone group), and -55.3 (calcipotriol group).
- In the combination group, mean PASI at the end of the double-blind phase was 2.5, and 3.6 at the end of the unblinded phase compared with 3.9 and 4.1, respectively for betamethasone alone and 4.4 and 3.7 for calcipotriol.
- In the double-blind phase, 8.1% of patients (combination) reported lesional/perilesional adverse reactions compared to 4.7% (betamethasone alone) and 12.0% (calcipotriol).
The calcipotriol/betamethasone combination was more effective and had a more rapid onset of action than either active constituent used alone, and was well tolerated. It is safe to transfer patients from betamethasone/calcipotriol to calcipotriol alone, with the maintenance of clinical effect.
Calcipotriol/betamethasone dipropionate ointment in elderly patients
- A total of 1534 patients participating in four randomised double-blind studies included 357 patients aged ≥ 60 years with psoriasis that received the two-compound calcipotriol/betamethasone dipropionate ointment once-daily [7].
- After 4 weeks of treatment, the mean reduction in PASI was 67.8% in patients < 60 years compared with 72.6% in patients ≥ 60 years.
- ‘Controlled disease’ based on a global assessment of ‘absence of disease’ or ‘very mild disease’ according to the investigators, was achieved by 52.1% of patients < 60 years and 58.2% of patients ≥ 60 years.
- Patients in both age groups reported a similar number of lesional/perilesional adverse drug reactions; 6.4% in patients < 60 years vs 5.0% in patients ≥ 60 years confirming that the two-compound ointment is effective and well-tolerated in the treatment of plaque psoriasis, regardless of age.
Cost-effective analyses
A cost-utility analysis from a Swedish healthcare payer’s perspective used a decision-tree model with a 12-week time horizon to evaluate the cost-effectiveness of calcipotriol/betamethasone foam versus calcipotriol/betamethasone ointment [8].
- 376 patients with plaque psoriasis received treatment with two 4-week cycles of topical calcipotriol 50 μg/g and betamethasone 0.5 mg/g as dipropionate (Cal/BD) foam or Cal/BD ointment as required before progressing to second-line treatment with phototherapy/methotrexate.
- The superior efficacy of Cal/BD foam over ointment led to fewer consultation visits, a decreased risk of progressing to second-line therapy, and lower total costs.
- Although Cal/BD foam costs more than Cal/BD ointment, this was offset by lower costs for phototherapy/methotrexate and fewer consultation visits.
Summary
- The two-compound ointment with calcipotriol 50 μg/g and betamethasone dipropionate 0.5 mg/g should be seen as the first-line treatment of choice for chronic plaque psoriasis amenable to topical treatment
- The product is a once-daily formulation and provides faster and greater symptom relief than a potent topical steroid used alone.
- Its formulation works in two ways to reduce inflammation and to treat the scaling associated with psoriasis
- This product is fully funded in New Zealand. Note that the foam formulation is not yet funded in New Zealand (January 2020).
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).