What is dupilumab?
Dupilumab is an innovative first-in-class biological treatment for atopic dermatitis (eczema).
Dupilumab (Dupixent®; Sanofi, Paris, France; Regeneron, New York, USA) is a fully human monoclonal antibody, which has shown significant efficacy and a favourable safety profile in moderate-to-severe atopic dermatitis alone and in combination with topical corticosteroids.
The U.S. Food and Drug Administration (FDA) approved Dupixent® as a treatment for moderate-to-severe atopic dermatitis in adults in March 2017, in patients aged 12–17 in March 2019, and in children age 6–11 in May 2020. It was approved by Therapeutic Goods Administration (TGA) in Australia in January 2018, and it was classified as a prescription medicine by Medsafe in New Zealand in March 2018.
The Biologics License Application (BLA) for dupilumab contains data from three pivotal phase 3 clinical studies evaluating dupilumab as monotherapy and in concomitant administration with topical corticosteroids.
What is dupilumab used for?
- Dupilumab is indicated for the treatment of patients over the age of 12 years with inadequately controlled moderate to severe atopic dermatitis.
- Current treatment options include moisturisers, antihistamines, topical corticosteroids or topical calcineurin inhibitors (TCIs), systemic corticosteroids, systemic calcineurin inhibitors, phototherapy, and other oral immune suppressants.
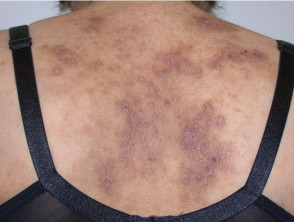
Severe atopic dermatitis in adults
How does dupilumab work?
- Dupilumab is thought to work by blocking the inflammation that causes atopic dermatitis.
- Atopic dermatitis is characterised by type 2 helper T (Th2) cell-driven inflammation,
- Dupilumab, a fully-human monoclonal antibody, is directed against the shared IL-4 receptor alpha subunit, which blocks signalling from both IL-4 and IL-13.
- IL-4 and IL-13 are key cytokines (signalling molecules that are made by cells and help to control the immune system and fight disease) that are required for the initiation and maintenance of the Th2 (Type 2 helper T-cell) immune response, which is believed to be a critical pathway in allergic inflammation.
Dosage and administration
- Dupilumab is administered as a subcutaneous injection.
- Dupilumab is intended as a long-term treatment.
- If discontinued for any reason, it can be restarted.
There is some evidence supporting successful interval extension between maintenance doses.
Link to key clinical-trial evidence about dupilumab.
What are the adverse effects of dupilumab?
Most common (> 5%) adverse reactions associated with dupilumab treatment in clinical trials were:
- Injection site reactions
- Ocular adverse effects (see below)
- Herpes infections
- Atopic dermatitis exacerbation
- Nasopharyngitis
- Headache
- Upper respiratory tract infection
No drug-specific blood-test monitoring is required.
Rarely, induction of another skin disorder has been reported, such as psoriasis.
Cutaneous T-cell lymphoma has been reported in 27 persons who recieved dupilumab. It may be that effective control of eczema has unmasked an underlying lymphoma, or that the original eruption was misdiagnosed as eczema. Affected individuals have been older (50-60 years), with extensive eczema (surface area exceeding 50%). Clues included failure of response or progression of the eruption despite dupilumab therapy, and the morphology of the eruption; these features should prompt consideration of biopsy.
Ocular adverse effects
Adverse effects involving the eye are wide-ranging in severity and frequency. Some studies suggest that dupilumab-associated ocular surface disease (DAOSD) may be greater in those with severe atopic dermatitis. These include:
- Conjunctivitis (in up to 30%)
- Dry eyes
- Allergic conjunctivitis
- Eye pruritus
- Other occular surface disease eg, limbal nodules
Treatment of conjunctivitis
Mild conjunctivitis is managed with lubricating eye drops. Patients on dupilumab should be advised to use these from the onset of treatment to prevent ocular symptoms. Some patients may require other treatment.
- Topical corticosteroid drops
- Topical tacrolimus eyelid ointment
- Referral to an ophthalmologist
Dupilumab is promising for atopic dermatitis in phase 3 studies
- Dupilumab is a fully human monoclonal antibody that inhibits the actions of both IL-4 and IL-13.
- Clinical trials of systemic dupilumab in moderate-to-severe atopic dermatitis have demonstrated a marked improvement in patient symptoms, including pruritus and clinically visible disease.
- Importantly, dupilumab treatment has been correlated with changes in the molecular signature of diseased skin, with reduction of both inflammatory and proliferative markers.
- Topical corticosteroids, emollients, phototherapy and systemic agents (such as prednisone, ciclosporin, methotrexate or azathioprine) can be continued during treatment with dupilumab. The dose of systemic agents may need to be reduced and they can be weaned off as the dupilumab begins to control the dermatitis.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).