Squamous cell carcinoma (SCC) is common form of keratinocytic skin cancer, usually related to exposure to ultraviolet radiation from sunlight. It often arises within solar/actinic keratosis or within squamous cell carcinoma in situ.
Histology of SCC
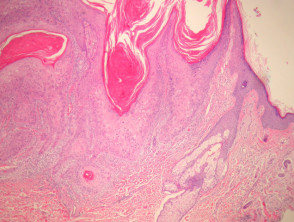
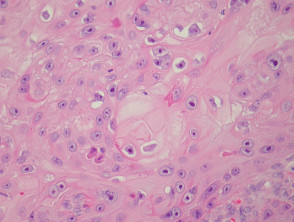
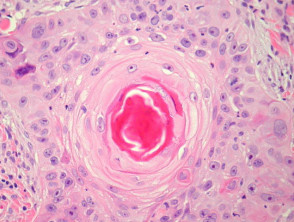
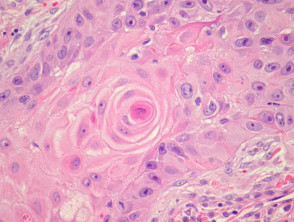
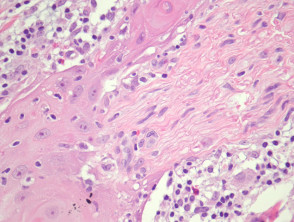
Typical SCC has nests of squamous epithelial cells arising from the epidermis and extending into the dermis (figure 1). The malignant cells are often large with abundant eosinophilic cytoplasm and a large, often vesicular, nucleus. Variable keratinisation (keratin pearls etc) is present (figure 2).
Squamous cell carcinoma – typical features
Typically SCC is graded as:
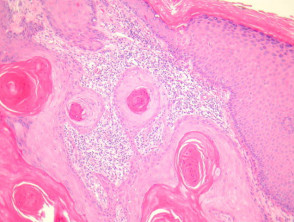
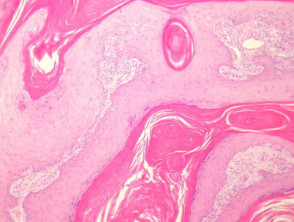
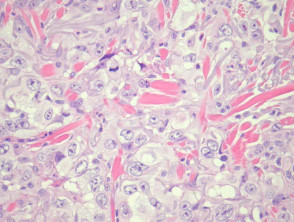
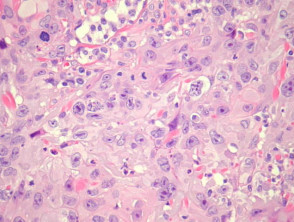
- Well differentiated (figures 3, 4)
- Moderately differentiated (figures 5, 6)
- Poorly differentiated (figures 7, 8)
Grading of SCC depends on how easy it is to recognise the characteristics of squamous epithelium (eg. intracellular bridges, keratinisation), pleomorphism and mitotic activity. There is considerable inter-observer variation in grading SCC.
Squamous cell carcinoma – differentiation
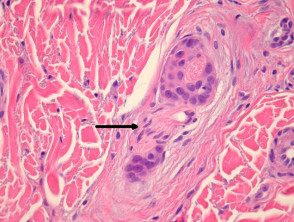
SCC can infiltrate along nerve sheaths, adventitia of blood vessels and lymphatics. Inflammation around these structures may be a clue to tumour in these areas. Tumour cells may evoke a stromal desmoplastic response. Extra-tumoural vascular invasion and neural invasion may be seen (figures 9, small – arrow indicates nerve; 10, large).
Squamous cell carcinoma – neural invasion
Variants of SCC
Special stains in SCC
Immunoperoxidase staining may be helpful in poorly differentiated and spindle cell-type SCC. EMA, MNF116, cytokeratin5/6 and p63 are expressed in SCC.
Differential diagnosis of SCC
SCC is usually not difficult to recognize pathologically. Deciding if the lesion is invasive or in situ can be more difficult, requiring levels through the specimen to be examined. Benign epithelial changes seen in the context of ulceration, stasis changes or infection can mimic SCC including:
- Reactive atypia
- Pseudoepitheliomatous hyperplasia
- Eccrine squamous syringometaplasia
- Viral changes
Other tumours may resemble SCC eg keratoacanthoma.
The differential diagnosis of SCC variants can be extensive.
- Clear cell SCC can resemble sebaceous carcinoma or balloon cell melanoma, etc.
- Acantholytic SCC may resemble angiosarcoma or adenocarcinoma. Clues these variants are SCC are often found in other parts of the lesion.
- Spindle cell SCC often requires immunohistochemistry for distinction from differential diagnoses including: leiomyosarcoma, angiosarcoma, melanoma, undifferentiated sarcoma, atypical fibroxanthoma, nodular fasciitis, Kaposi sarcoma, dermatofibrosarcoma protruberans.