Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Chemical burn is a burn to internal or external organs of the body caused by a corrosive or caustic chemical substance that is a strong acid or base (also known as alkali). Chemical burns are usually the result of an accident and can occur in the home, at school or more commonly, at work, particularly in manufacturing plants that use large quantities of chemicals.
Very mild chemical burns result in irritant contact dermatitis. Chemical burn from a strong acid or alkali is also known as a caustic burn.
The main cause of chemical burn is contact with strong acids or bases.
Sulphuric acid – concentration ranges from 8% to almost pure acid
Nitric acid
Hydrofluoric acid – a weak acid and in a dilute form does not burn or cause pain on contact
Hydrochloric acid – concentrations range from 5–44%
Phosphoric acid
Sodium hydroxide and potassium hydroxide — depending on the concentration — may be very corrosive
Sodium and calcium hypochlorite
Ammonia
Phosphates
The signs and symptoms of a chemical burn depend on several factors, including:
Swallowing a solid pellet of an alkaline substance highlights the importance of these factors. The solid pellet sits in the stomach for a longer period, thus more severe burns sustained. Another important factor is concentrated forms of some acids and bases generate a large amount of heat when diluted; this results in a thermal burn as well as a chemical burn.
Some signs and symptoms of chemical burns include:
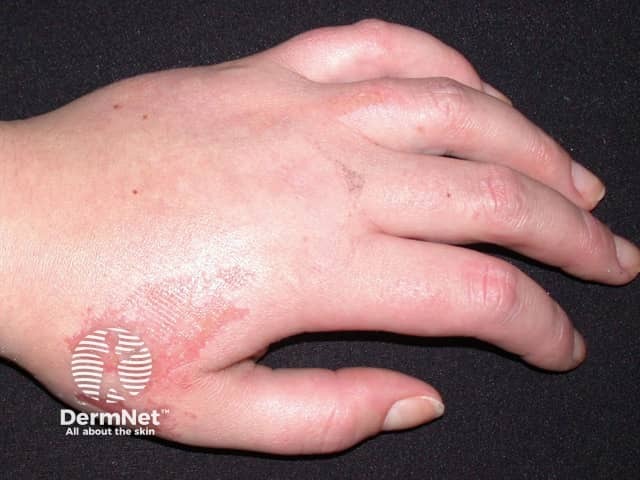
Unknown cause
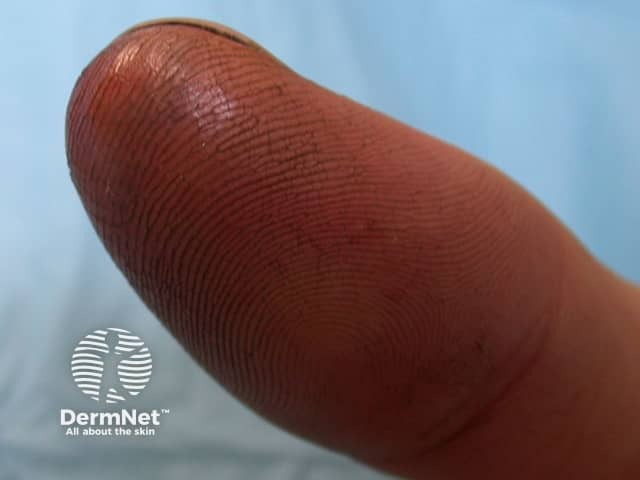
24 hours after fluoride burn

10 days after fluoride burn
In severe chemical burns where the agent has been swallowed, inhaled or absorbed into the bloodstream, the following systemic symptoms may occur.
Basic first aid should be administered as soon as a chemical burn has occurred.
Chemical burns involving elemental metals (lithium, potassium, sodium and magnesium) should not be irrigated with water as this can result in a chemical reaction that causes burns to worsen. These types of chemical burn should be soaked with mineral oil while waiting for medical attention.
People with minor chemical burns do not require hospitalisation. For more severe burns, patients should receive treatment as for a typical thermal burn patient. In some situations an antidote may be given to counteract the offending chemical agent. For example, hydrofluoric acid burns should be promptly treated with calcium gluconate gel applied every 15 minutes, so the gel should be kept at relevant work sites.
The main treatment aims of burn wound management are:
Commonly used topical antibacterials include 1% silver sulfadiazine cream, 0.5% silver nitrate solution and mafenide acetate 10% cream.