What is benzocaine?
Benzocaine (chemical formula C9H11NO2) is a widely used local anaesthetic. It is classified as an ester type of local anaesthetic (as opposed to an amide), due to the para-aminobenzoic acid in its structure.
Benzocaine is frequently linked to hypersensitivity reactions. The prevalence of these reactions is thought to be due to the presence of benzocaine in many over-the-counter (OTC) products. The greater the exposure to a potential allergen, the more likely sensitisation is to occur.
Where is benzocaine found?
Benzocaine is used in pharmaceutical preparations and, rarely, in cosmetics. It is mainly found in gel and liquid formulations. These are available as OTC medications to suppress coughs, or to treat pain due to mouth ulcers, pharyngitis, or haemorrhoids.
Preparations containing benzocaine include:
- Wound and burn preparations
- Sunburn remedies
- Haemorrhoid preparations
- Oral and gingival products
- Sore throat sprays and lozenges
- Callus and wart remedies
- Creams for treatment of poison ivy
- Toothache and denture irritation products.
Doctors and dentists use benzocaine preparations on mucosal surfaces such as the mouth, to prepare or 'numb' a site for injection; it is not injected.
What are the clinical features of a benzocaine allergy?
Allergies to local anaesthetic are nearly always a delayed-type hypersensitivity reaction [1]. This requires sensitisation; a reaction will occur approximately 12–24 hours after re-exposure to the allergen.
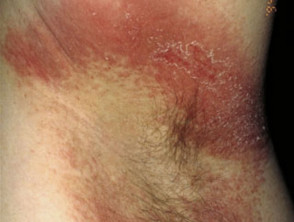
Benzocaine sensitivity produces a classic allergic contact dermatitis in the area of topical application. This manifests as acute erythema, and in severe cases blistering and bullae where it has been in contact with the skin. Sometimes, benzocaine applied to an existing rash may cause it to flare or spread.
Occasionally, the injection of another PABA-derived local anaesthetic to a benzocaine-allergic individual may cause swelling of the oral mucosa at the site of the injection. This may appear similar to an immunoglobulin E (IgE) mediated urticarial reaction; however, the timeline and absence of respiratory compromise indicate a cell-mediated response.
The incidence of IgE-mediated reactions to all local anaesthetics is < 1% [2]. There are very few reports in the literature of anaphylaxis caused by any type of local anaesthetic.
Contact allergy to benzocaine
How is benzocaine allergy diagnosed?
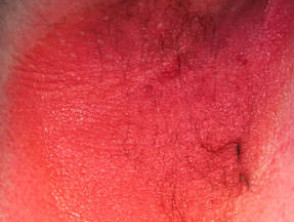
If benzocaine contact allergy is suspected based on the patient's history and examination, patch testing is performed to confirm the hypersensitivity reaction, using 5% benzocaine in petrolatum [3]. Other local anaesthetics will also usually be tested, as cross-reactivity between ester local anaesthetics is common because of the structural similarities.
Positive patch test to benzocaine
What is the treatment of benzocaine allergy?
The treatment of hypersensitivity to benzocaine is to treat the acute rash and to avoid benzocaine-containing products.
The acute dermatitis treatment is as for any acute eczema, such as topical corticosteroids, emollients, and treatment of any secondary bacterial infection, such as Staphylococcus aureus.
To avoid benzocaine-containing products, product labels must be carefully inspected for benzocaine or any of its alternative names. Any products that are labelled 'anaesthetic' or 'caine' should be suspected of containing benzocaine or a related compound.
Other PABA-based products should also be avoided because of the risk of cross-reactivity, including:
- PABA, meta-aminobenzoic acid, or benzoic acid local anaesthetics (both topical and injectable forms)
- Paraphenylenediamine found in permanent hair dyes (see hair dye allergy)
- Sulfonamides
- Sulfonylureas
- PABA-based sunscreens
- Thiazide-related diuretics.
An amide local anaesthetic can usually be used safely in a benzocaine-allergic patient, as they do not contain PABA.
What other complications are associated with benzocaine use?
Benzocaine products have been linked to the development of methaemoglobinaemia. Methaemoglobin is an oxidised form of haemoglobin that cannot readily release oxygen, shifting the oxygen-dissociation curve to the left and resulting in hypoxia. Methaemoglobinaemia from benzocaine has been reported when a benzocaine-containing spray was used during bronchoscopy (a procedure to examine the airways with a fiberoptic scope) or gastroscopy (a procedure to examine the interior of the stomach with a gastroscope) [4].