What is azathioprine and mercaptopurine?
Azathioprine is an antimetabolite thiopurine analogue drug that interferes with DNA synthesis and suppresses the immune system response.
Azathioprine is metabolised in the liver to mercaptopurine, also known as 6-mercaptopurine (6-MP) which is also commercially available as a drug. Mercaptopurine is then converted into thioguanine nucleotides which inhibit cell growth.
Who uses azathioprine and mercaptopurine?
Azathioprine is more often used in practice than mercaptopurine for the treatment of skin conditions, however they are closely related.
Approved azathioprine indications may include:
- Prevention of organ transplant rejection, rheumatoid arthritis, Crohn disease
- Dermatology-approved indications in some countries include: systemic lupus erythematosus, dermatomyositis, polyarteritis nodosa.
Azathioprine may be approved to treat these skin conditions
Off-label uses in dermatology with good evidence include:
- Atopic dermatitis
- Behçet disease
- Bullous pemphigoid
- Cutaneous polyarteritis nodosa
- Pemphigus vulgaris
- Prurigo
- Psoriasis
- Pyoderma gangrenosum
- Relapsing polychondritis
- Sarcoidosis
Limited evidence is available for many other dermatologic conditions.
What are the contraindications and precautions with azathioprine and mercaptopurine?
Contraindications
- Drug or excipient allergy
- Pregnancy (relative contraindication — see below)
- Very low thiopurine methyltransferase (TPMT) activity (relative contraindication — see below)
- Concomitant use of allopurinol or febuxostat (relative contraindication)
Precautions
- Live vaccines [see Immunisation in immunosuppressed dermatology patients]
- Phototherapy due to skin cancer risk
Pregnancy and breastfeeding
Treatment with azathioprine and mercaptopurine during all stages of pregnancy should be avoided except where benefits outweigh risks.
Females of childbearing potential should be advised of the risks involved and use effective contraception where appropriate. Teratogenicity is unclear in men taking azathioprine or mercaptopurine and adequate contraception may also be advised in this case.
Although low concentrations of 6-mercaptopurine may be found in breastmilk, there has been no evidence to suggest harm. Potential benefits should outweigh risks; for further information, see Lactation and medications used in dermatology.
Tell me more about azathioprine and mercaptopurine
Mechanism of action
Azathioprine has been widely prescribed since the 1960s, however its mechanism of action as well as that of its metabolite 6-MP is not fully understood. There are a number of proposed mechanisms including:
- Reduction in cell division through self-incorporation into DNA and RNA as a false nucleotide
- Immunosuppression occurs after mercaptopurine crosses cell membranes and becomes intracellularly activated.
Drug forms and dosing
Azathioprine and mercaptopurine may be available as an oral tablet, oral liquid, or intravenous injection (azathioprine only).
The dose of azathioprine is generally 1–3 mg/kg/day, however the individualised dose is dependent on several factors including indication, age, response, and TPMT activity (see below). The bioavailability may vary between different formulations. Dose reduction may be required in patients with hepatic and renal impairment.
Effects of azathioprine and mercaptopurine may take up to several months to be seen — this should be considered when reviewing treatment.
What are the benefits of azathioprine and mercaptopurine?
- Potential steroid-sparing effect
- Multiple dosage forms and strengths, allowing for easy dose titration
- Oral formulation available
What are the disadvantages of azathioprine and mercaptopurine?
- May take several months for the full benefits to be seen
- Metabolism varies between individuals (see TPMT monitoring below)
- Regular blood test monitoring is required
What are the side effects and risks of azathioprine and mercaptopurine?
Side effects
Azathioprine and mercaptopurine usually cause mild side effects but may occasionally be severe enough to stop treatment. These include:
- Mucocutaneous side effects
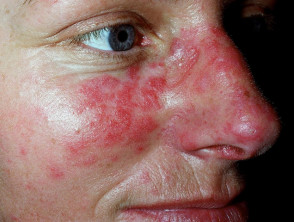
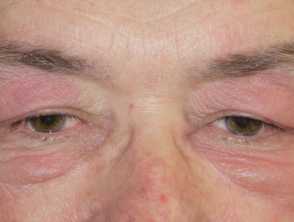
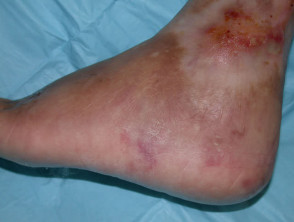
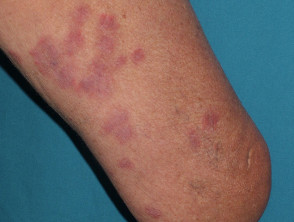
- photosensitivity, hair loss, skin infections, acute febrile neutrophilic dermatosis, acne, and pellagra
- cutaneous squamous cell carcinoma with long term treatment over years
- Stevens-Johnson syndrome/toxic epidermal necrolysis and drug hypersensitivity syndrome - rare but serious side effects
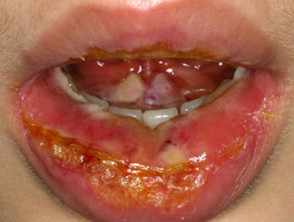
Mucocutaneous side effects that may be caused by azathioprine
- Common side effects
- nausea and dose-related bone marrow suppression
- Less common side effects
- diarrhoea, vomiting, infections, liver impairment, and pancreatitis.
Drug interactions
If the use of an interacting drug combination is unavoidable, dose adjustment may be required and blood counts should be monitored carefully. Interactions include:
- Warfarin dose requirement may increase 3-4 fold when given with azathioprine [see Anticoagulants and antiplatelet agents]
- Trimethoprim + sulfamethoxazole
- Angiotensin-converting enzymes inhibitors
- Neuromuscular blocking agents eg, rocuronium, atracurium
- Indomethacin
- Sulfasalazine and other benzoic acid derivatives
Monitoring during treatment
Before starting treatment:
- Testing for latent tuberculosis, hepatitis B and C
- Checking immunity status eg, measles and varicella [see Immunisation in immunosuppressed dermatology patients]
- Baseline blood tests and skin check.
During treatment:
- Regular full blood counts are checked weekly at first for the first 4–8 weeks, then at longer intervals.
- Liver and kidney function tests — baseline and periodically
- Regular skin checks — patients should practice self skin examination.
Thiopurine methyltransferase (TPMT)
Azathioprine and mercaptopurine is primarily metabolised by the enzyme TPMT. Some people have a genetic mutation causing them to have either low enzyme activity (approximately 11% of the population) or a lack of enzyme activity (1 in 300 people). Those with both genes are at severe risk of bone marrow suppression. Conversely, some other individuals have high levels of enzyme activity and may require a higher dose than normal.
TPMT levels should be measured to determine a patient's level of risk before starting treatment. Low levels are < 5 U/mL, intermediate levels are 5–13.7 U/mL, and high levels are > 13.8 U/mL.
Discontinuation of treatment
Azathioprine should be stopped and promptly managed jointly with a haematologist if:
- Neutrophil count < 1.0x109/L
- Lymphocyte count < 0.5x109/L
- Platelet count < 50x109/L.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).