Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Treatments Lesions (cancerous)
Author: Anoma Ranaweera, Medical Writer, Auckland, New Zealand. DermNet Chief Editor in Chief: Adjunct A/Prof Dr Amanda Oakley, Dermatologist, Hamilton, New Zealand. First published December 2014; updated March 2018. Copy edited by Gus Mitchell/Maria McGivern.
This article was updated with financial support from Merck Sharp & Dohme (New Zealand) Limited, distributors of Keytruda® in New Zealand. Sponsorship does not influence content.
Introduction
Benefits in melanoma patients
How it works
How to use
Potential drug interactions
Adverse effects
Use in particular populations
Pembrolizumab (formerly known as lambrolizumab; trade name Keytruda®) is a drug marketed by Merck and Co (New Jersey, USA) that targets the programmed cell death 1 (PD-1) receptor. The drug is intended for use in treating metastatic melanoma.
On 4 September, 2014, the US Food and Drug Administration (FDA) approved pembrolizumab as a breakthrough therapy for the treatment of metastatic melanoma. It was approved for use for advanced melanoma in New Zealand in September 2015 and was funded by the NZ Pharmaceutical Management Agency (PHARMAC) for some patients with unresectable or metastatic melanoma from September 2016. Pembrolizumab is also used for metastatic non-small cell lung carcinoma, some cases of Hodgkin lymphoma, urothelial carcinoma (March 2018) and cutaneous squamous cell carcinoma (FDA, June 2020). Pembrolizumab is under investigation in other malignancies. It has been reported to have some effect in metastatic Merkel cell carcinoma.
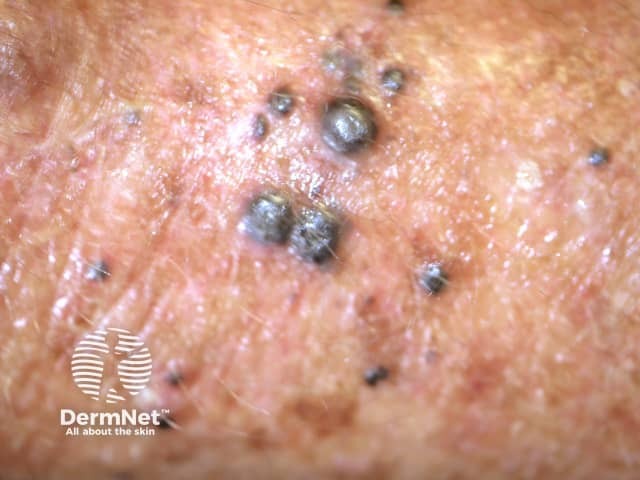
Cutaneous melanoma
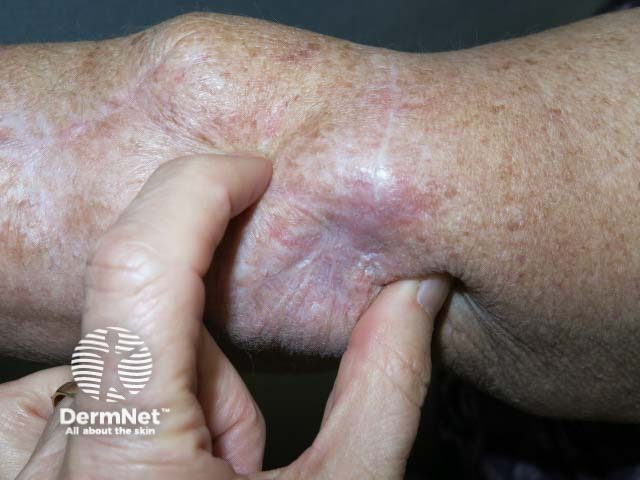
Cutaneous melanoma mestastases
Cutaneous melanoma mestastases
Pembrolizumab is indicated for the treatment of patients with unresectable or metastatic melanoma.
See key clinical-trial evidence about pembrolizumab.
Combination therapy with other immunological drugs and targeted cancer therapy is under investigation and may improve response rates.
The programmed death receptor (PD-1) is a surface molecule expressed on antigen-stimulated T cells as well as monocytes, B cells, and dendritic cells. In normal cells, the PD-1 receptor acts as an immune checkpoint receptor enabling self-tolerance by T cells, thus preventing autoimmune reactions.
When unbound, PD-1 allows the normal response by T-cells to proceed. However, the binding of PD-1 to its ligands, programmed death receptor ligand 1 (PD-L1) and programmed cell death ligand 2 (PD-L2), suppresses the immune response by inducing downstream signalling that inhibits the proliferation of T cells, cytokine release, and cytotoxicity.
Abnormal PD-L1 expression on the surface of melanoma cells activates PD-1 and suppresses cytotoxic T-cell activity. This T-cell tolerance enables the tumour cells to avoid recognition and attack by the immune system.
Pembrolizumab is a highly selective humanised monoclonal immunoglobulin G4 (IgG4) antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing the PD-1 pathway-mediated inhibition of the immune response against tumour cells.
No formal pharmacokinetic drug interaction studies have been conducted with pembrolizumab.
In clinical trials, the most common side effects (≥ 20%) in patients receiving pembrolizumab 2 mg/kg every 3 weeks were:
The most frequent (≥ 2%) serious adverse drug reactions observed with pembrolizumab were renal failure, dyspnoea (breathlessness), pneumonia, and cellulitis. Pembrolizumab can also cause infusion reactions including anaphylaxis.
Pembrolizumab also has the potential for severe immune-mediated adverse effects. In 411 clinical trial participants with advanced melanoma treated with pembrolizumab, severe immune-mediated adverse effects included pneumonitis, colitis, hypophysitis (inflammation of pituitary gland), hyperthyroidism, hypothyroidism and other endocrinopathies, nephritis, and hepatitis. Other autoimmune diseases have subsequently been reported in association with pembrolizumab.
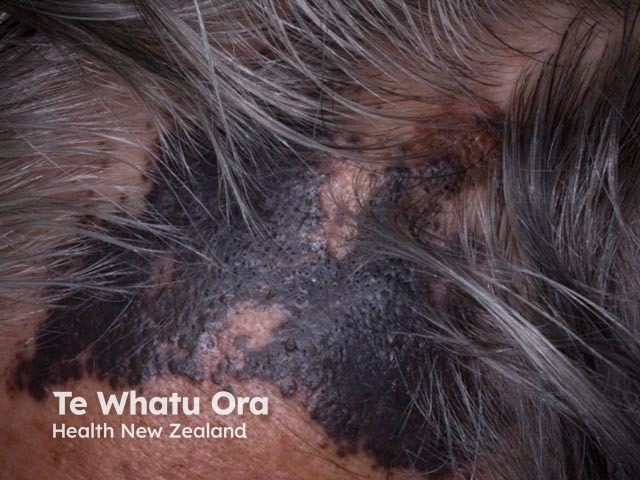
One report has described cutaneous adverse effects arising in 35 of 83 (42%) patients treated with pembrolizumab. The development of skin problems was associated with longer progression-free intervals. Case reports have described other skin problems during the drug's post-marketing period; these skin problems have included:
The darkening of hair has been reported in a few patients taking PD-1 inhibitors for non-small cell lung cancer.
See also Cutaneous adverse effects of checkpoint inhibitors.
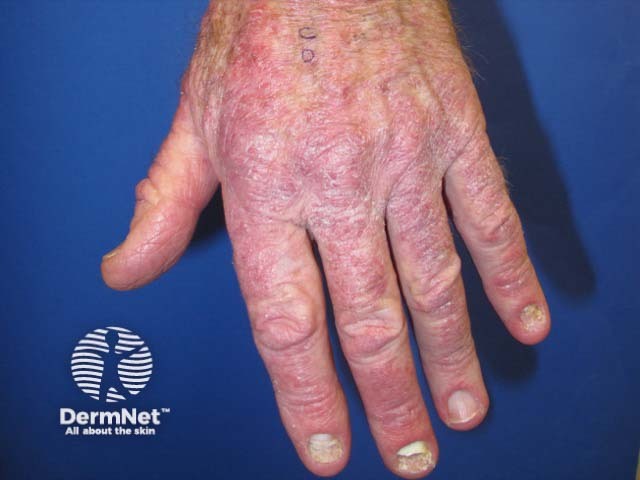
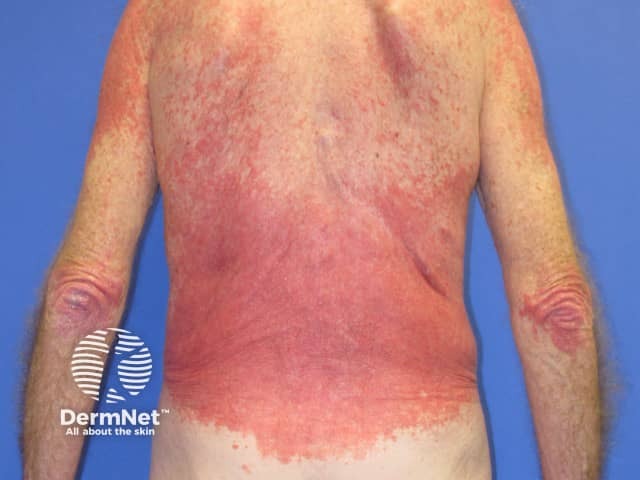
Pembrolizumab-induced psoriasis
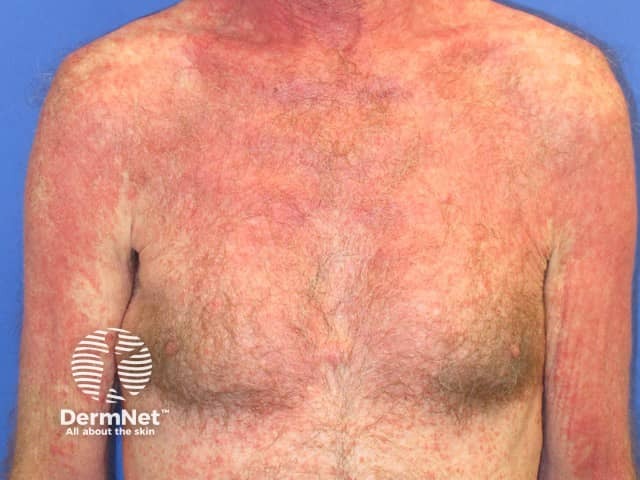
Pembrolizumab-induced psoriasis
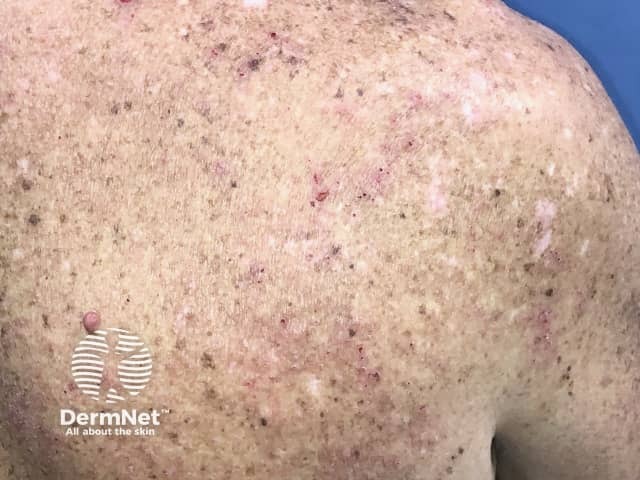
Eczema induced by pembrolizumab
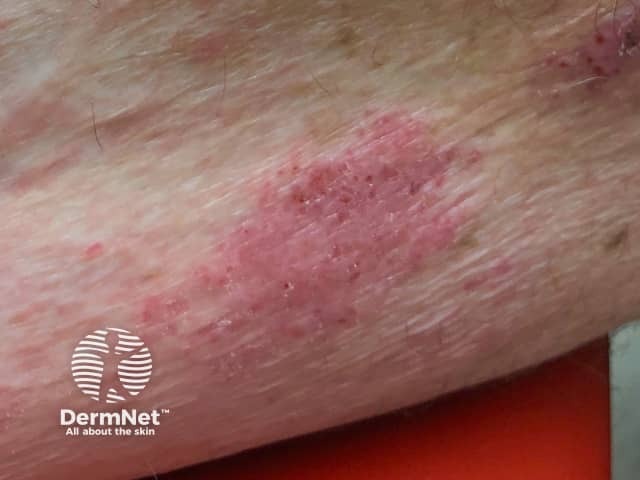
Eczema induced by pembrolizumab
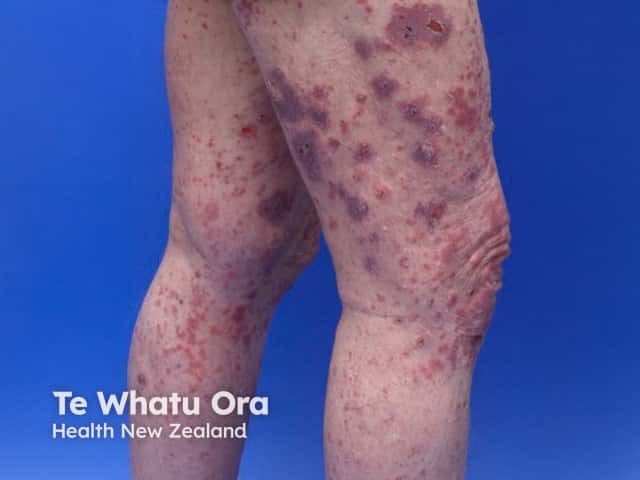
Bullous pemphigoid induced by pembrolizumab
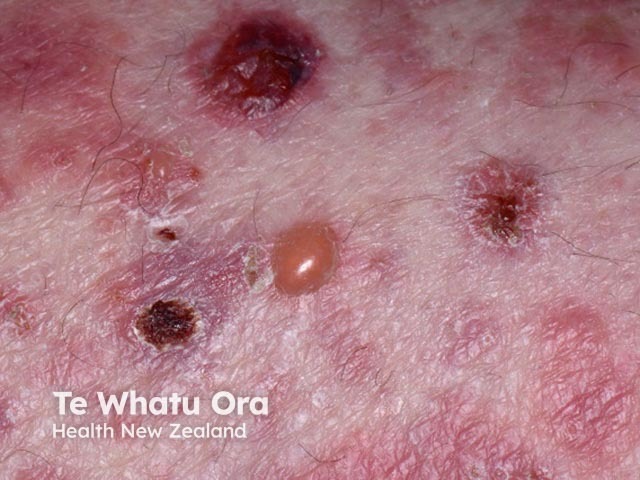
Bullous pemphigoid induced by pembrolizumab
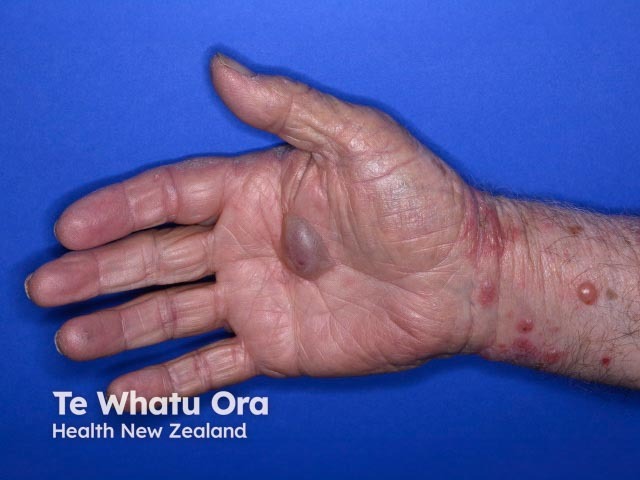
Bullous pemphigoid induced by pembrolizumab
It is not known whether pembrolizumab or its metabolites are excreted in human milk. Because many drugs are excreted in human milk, mothers should discontinue nursing prior to using pembrolizumab.
The safety and effectiveness of pembrolizumab have not been established in children.
In one trial of 411 clinical trial patients treated with pembrolizumab, 39% were 65 years and over. No overall differences in safety or efficacy were reported between older patients and younger patients.
Based on a population pharmacokinetic analysis, no dose adjustment is needed for patients with renal impairment.
Based on a population pharmacokinetic analysis, no dose adjustment is needed for patients with mild hepatic impairment. Pembrolizumab has not been studied in patients with moderate or severe hepatic impairment.
Approved datasheets are the official source of information for medicines, including approved uses, doses, and safety information. Check the individual datasheet in your country for information about medicines.
We suggest you refer to your national drug approval agency such as the Australian Therapeutic Goods Administration (TGA), US Food and Drug Administration (FDA), UK Medicines and Healthcare products regulatory agency (MHRA) / emc, and NZ Medsafe, or a national or state-approved formulary eg, the New Zealand Formulary (NZF) and New Zealand Formulary for Children (NZFC) and the British National Formulary (BNF) and British National Formulary for Children (BNFC).