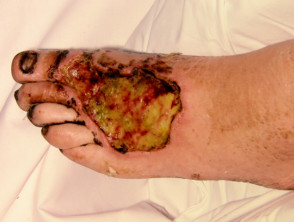
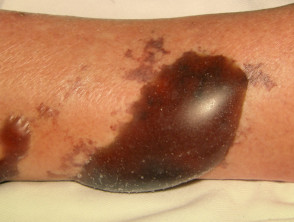
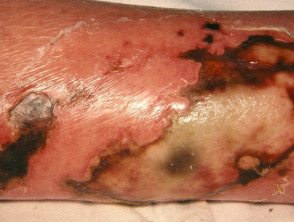
What is wet gangrene?
Gangrene the localised death of body tissue. Wet gangrene is gangrene due to necrotising bacterial infections, including necrotising fasciitis. Wet gangrene should be distinguished from 'dry' gangrene, which is due to ischaemia.
What causes necrotising bacterial infections?
Necrotising bacterial infections can occur via any break in the skin or internal organ. They are caused by 3 main bacterial subgroups:
1. Polymicrobial necrotising infection
Polymicrobial necrotising infections often involve a mix of:
- gram-positive cocci
- gram-negative rods
- anaerobes, including clostridial species.
Polymicrobial necrotising infections tend to affect the trunk and perineum.
Patients with these infections usually have a history of other medical problems, especially diabetes, and are likely to be older adults. The initial injury to the skin may have been unnoticed.
Gas gangrene
Gas gangrene is particularly severe and is most often due to Clostridium perfringens, which can rapidly proliferate in injured muscles. This organism is ubiquitous in soil and dust. Gas gangrene was very prevalent in World War 1, complicating 6% of open fractures and 1% of all open wounds. Clostridia release alpha, beta and other toxins, which induce blood clotting at the site of the infection and result in myonecrosis (death of muscle tissue). Reduced blood flow, localised ischaemia and lowered pH result in a favourable environment for other bacteria to grow. The gas is composed of nitrogen, oxygen, hydrogen, hydrogen sulfide and carbon dioxide. It spreads within the muscle fibres allowing rapid spread of infection.
2. Group A streptococcus and staphylococcus
Group A β-hemolytic streptococci (GAS), either alone or associated with staphylococcal species, tend to be locally aggressive and can lead to serious blood infections such as toxic shock syndrome.
Patients with streptococcal and staphylococcal infections are likely to be younger and in better general health than those with polymicrobial infections. The route of entry usually follows trauma, including surgery and intravenous drug (ab)use. M proteins produced by GAS allow the bacteria to adhere to tissue and evade the immune system. M protein can also activate T lymphocytes, which can lead to an inflammatory shock response.
3. Gram-negative marine organisms
Marine organisms such as Vibrio vulnificus are rare causes of gangrene and have mostly been reported in warm coastal regions. Route of infection can be through an open wound exposed to water, or via ingestion of infected oysters. Systemic toxicity tends to occur early.
Who gets necrotising bacterial infections?
Risk factors for necrotising infections include:
- Diabetes mellitus
- Atherosclerosis and peripheral vascular disease
- Trauma
- Surgery, especially operations on colon or gallbladder
- Intravenous drug and insulin injection
- Skin infections, erosions and ulcers
- Animal and insect bites
- Visceral-cutaneous fistulas
- Percutaneous catheter insertion
- Abscesses
- Urinary tract infection
- Streptococcal sore throat (tonsillitis)
- Alcohol abuse
- Immune deficiency, including human immunodeficiency virus (HIV) infection
- Immune suppression by medications
- Anticoagulants, especially the combination of warfarin (coumadin) and heparin.
What are the clinical features of necrotising bacterial infections?
The clinical features of necrotising infections depend upon the location and cause.
Necrotising infections frequently resemble cellulitis or an abscess initially, but progress to discolouration (blue to black), foul-smelling discharge, and/or numbness. If the affected area is internal, symptoms may include confusion, fever, gas in the tissues under the skin, low blood pressure and persistent or severe pain.
The early signs of gangrene that should result in an urgent investigation are:
- Bullae (blisters), often haemorrhagic
- Ecchymosis (bruising) preceding necrosis (death of tissue)
- Crepitation (gas in the tissues)
- Cutaneous anaesthesia (numbness).
Less specific signs that should increase clinical suspicion of necrotising infection include:
- Pain out of proportion to the signs of disease
- Swelling that extends beyond the area of reddened skin
- Systemic toxicity
- Progression of infection despite treatment with antibiotics.
Necrotising skin infections may progress rapidly to wet gangrene, unlike cellulitis or abscess.
Necrotising skin infections with gangrene
Investigations in necrotising infections
Laboratory tests
Blood cultures should be undertaken to identify specific organisms involved in the infection.
The table below was developed as a risk indicator for necrotising fasciitis by Wong et al. in 2004. Scores ≥6 were found to have a 92% positive predictive value and a 96% negative predictive value.
| Laboratory marker | Scoring evaluation |
|---|---|
| CRP (mg/dL) | < 150 = 0 points > 150 = 4 points |
| WBC (per mm3) | < 15 = 0 points 15–25 = 1 point > 25 = 2 points |
| Hb (g/dL) | > 13.5 = 0 points 11.6–13.5 = 1 point < 11.5 = 2 points |
| Serum sodium | > 135 = 0 points < 135 = 2 points |
| Serum creatinine | ≤ 1.6 = 0 points > 1.6 = 2 points |
| Serum glucose | < 180 = 0 points > 180 = 1 point |
CRP: C-reactive protein; Hb: haemoglobin.
Imaging
Imaging has a limited role in the diagnosis of infectious gangrene.
- Plain film X-ray can demonstrate subcutaneous emphysema (gas under the skin) sometimes present in clostridial infections.
- CT scanning may show fascial thickening, oedema, subcutaneous gas, and abscess formation.
- MRI scanning shows similar findings to CT but is less specific as tissue enhancement may mimic other inflammatory processes.
Surgical exploration
- Surgery is the gold standard for diagnosing necrotising fasciitis — the fascia separates from surrounding tissue.
- Small incisions with local exploration are useful for identifying necrotic tissue in other cases.
What is the treatment for established wet gangrene?
Treatment of gangrene depends on location and cause, but is centred around:
- Radical surgical debridement +/- amputation
- Broad spectrum antimicrobial therapy – penicillin G, clindamycin, vancomycin, and gentamicin may be used for empiric therapy for methicillin-resistant Staphylococcus aureus (MRSA), gram-negative, gram-positive, and anaerobic organisms.
- Haemodynamic support.
Management may also include:
- Maggot debridement therapy — the maggots consume dead and infected tissue
- Vascular surgery to improve circulation
- Hyperbaric oxygen, especially in patients with diabetes or peripheral vascular disease.
- Intravenous immunoglobulin – this may limit the inflammatory response by binding staphylococcal and streptococcal toxins.
- Treatment of underlying conditions such as diabetes.
The incidence of wartime gas gangrene has markedly reduced with identification and isolation of cases, and early debridement, amputation and supportive treatment.
What is the prognosis for wet gangrene?
Prognosis in wet gangrene depends on the extent of disease, the underlying cause and the timing of appropriate treatment.
Anaya et.al have constructed a clinical score to predict patient outcomes for infective gangrene based on several clinical criteria:
- Heart rate > 110 beats per minute
- Temperature < 36 degrees Celsius
- Serum creatinine > 1.5 mg/dL
- Age > 50 years
- WBC > 40 000 per uL
- Haematocrit > 50%
Patients with few of these findings had a 6% risk of death in this study, while patients with several findings had up to an 88% risk of death. Prognosis can vary widely due to the aggressive nature of gangrene if treatment is not accessed early.