Main menu
Common skin conditions

NEWS
Join DermNet PRO
Read more
Quick links
Authors: Rachel Xuan, Medical Student, University of New South Wales, Sydney, NSW, Australia; Dr Anes Yang, Dermatology Clinical Research Fellow, Premier Specialists, Sydney, NSW, Australia; and Professor Dedee F. Murrell, Head, Department of Dermatology, St George Hospital, University of New South Wales, Sydney, NSW, Australia. DermNet Editor in Chief: Adjunct A/Prof Amanda Oakley, Dermatologist, Hamilton, New Zealand. Copy edited by Gus Mitchell. December 2017.
Introduction Conditions tested The laboratory method Interpreting results
Indirect immunofluorescence, or secondary immunofluorescence, is a technique used in laboratories to detect circulating autoantibodies in patient serum. It is used to diagnose autoimmune blistering diseases.
The tissue substrates used (whether monkey oesophagus or human skin) will depend on the suspected disease. Different staining patterns will help diagnose an autoimmune bullous disease.
Indirect immunofluorescence is used to diagnose the following autoimmune bullous diseases:
The immunofluorescent slides are examined to determine the presence of autoantibodies via the patterns of immune deposition. The results are subjective and indirect immunofluorescence cannot be used reliably to monitor disease severity and its treatment.
The different staining patterns seen with indirect immunofluorescence include the:
The sensitivity of a diagnostic tool refers to its ability to accurately identify those with the disease (ie, producing a true-positive result). The specificity of a diagnostic tool refers to its ability to accurately identify those without the disease (ie, producing a true-negative result).
Occasionally a test can also produce false results. Hence:
The immunofluorescent patterns seen in the following skin diseases are described below.
In pemphigus vulgaris, indirect immunofluorescence shows:
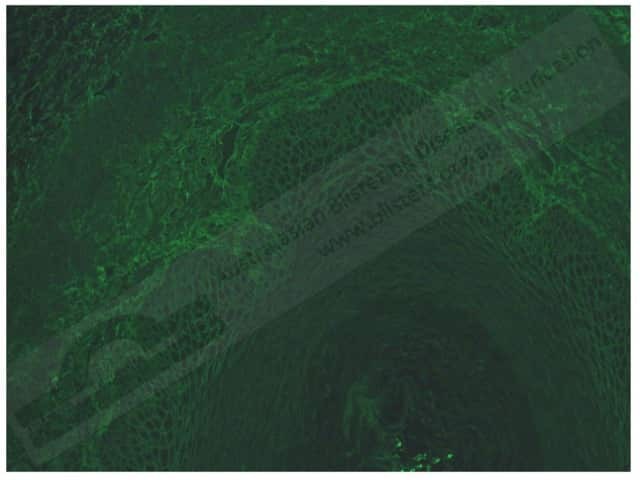
Pemphigus vulgaris
In pemphigus foliaceus, indirect immunofluorescence shows:
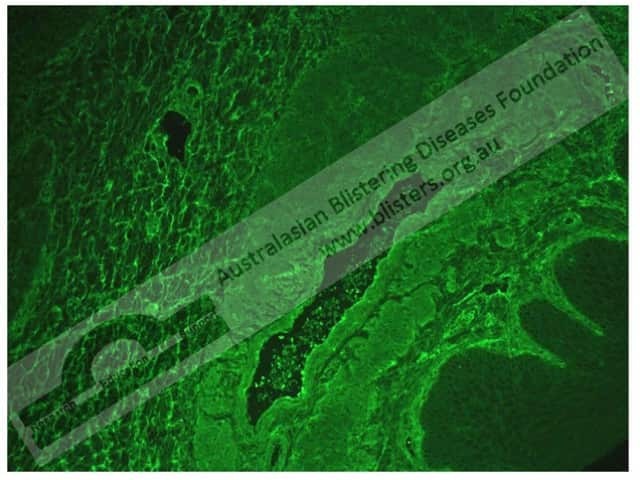
Pemphigus foliaceus
In pemphigus, indirect immunofluorescence shows:
In bullous pemphigoid, indirect immunofluorescence shows:
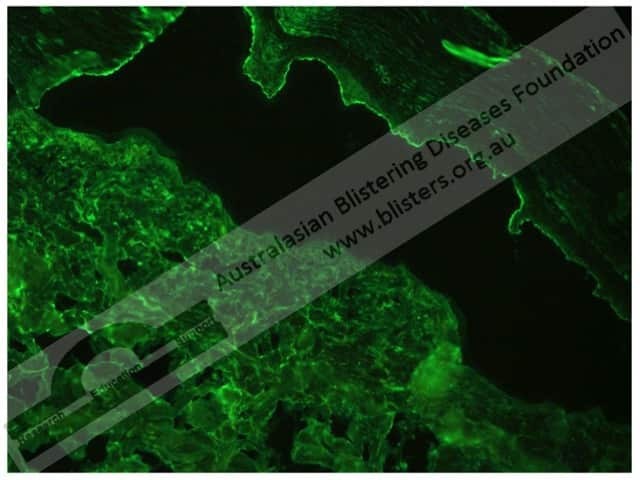
Bullous pemphigoid
In mucous membrane pemphigoid, indirect immunofluorescence shows:
In pemphigoid gestationis, indirect immunofluorescence shows:
In epidermolysis bullosa acquisita, indirect immunofluorescence shows:
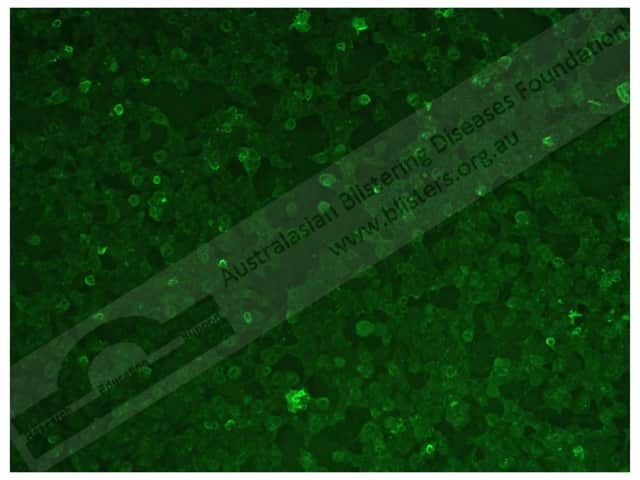
Epidermolysis bullosa acquisita
In dermatitis herpetiformis, indirect immunofluorescence shows:
In linear IgA bullous dermatosis and chronic bullous disease of childhood, indirect immunofluorescence shows:
In bullous systemic lupus erythematosus, indirect immunofluorescence shows a linear BMZ pattern with IgG antibodies.